| HYDRAZINE NITRATE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 13464-97-6 |
|
| EINECS NO. | 236-690-7 | |
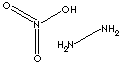
| FORMULA | H4N2·HNO3 |
|
| MOL WT. | 95.02 | |
|
H.S. CODE |
||
| TOXICITY | ||
| SYNONYMS | hydrazinium nitrate; Nitrate d'hydrazinium (French); | |
| Hydraziniumnitrat (Germana); Nitrato de hidrazinio (Spanish); | ||
| DESCRIPTION | ||
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES (50%) |
||
| PHYSICAL STATE | clear liquid | |
| MELTING POINT |
|
|
| BOILING POINT | ||
| SPECIFIC GRAVITY | 1.2 | |
| SOLUBILITY IN WATER | Soluble | |
| pH | 4 - 6 | |
| VAPOR DENSITY | ||
|
AUTOIGNITION |
||
|
NFPA RATINGS |
Health: 3; Flammability: 0; Reactivity: 0 | |
|
REFRACTIVE INDEX |
|
|
| FLASH POINT | ||
| STABILITY | Stable under ordinary conditions | |
|
GENERAL DESCRIPTION AND APPLICATIONS |
||
| Hydrazine (anhydrous), H2NNH2, is a clear, fuming, corrosive liquid with an ammonia-like odor; melting at 1.4 C, boiling at 113.5 C, specific gravity 1.011. It is very soluble in water and soluble in alcohol. It decomposes on heating or exposure to UV to form ammonia, hydrogen, and nitrogen, which may be explosive with a blue flame when catalysed by metal oxides and some metals such as platinum or Raney nickel. It is prepared from ammonia with chloramine in the presence of glue or gelatin (to inhibit decomposition of the hydrazine by unreacted oxidants) as the hydrate form usually (100% monohydrate contains 64% by weight hydrazine). Hydrazine is also prepared from sodium hypochlorite with urea in the presence of glue or gelatin. Botht ammonia and amines are nitrogen nucleophiles which donate electrons (they are Lewis bases). But hydrazine (diamine) has much stronger nucleophilicity which makes it more reactive than ammonia. Hydrazine has dibasic and very reactive properties. Hydrazine is used as a component in jet fuels because it produce a large amount of heat when burned. It is less flammable and less volatile than hydrocarbon fuels. It is relatively environmentally friendly because they degrade quickly in the environment. Hydrazine is used as an oxygen scavenger for water boiler feed and heating systems to prevent corrosion damage. Hydrazine is used as a reducing agent for the recovery of precious metals. It is used as a polymerization catalyst and a chain extender in urethane coatings. It, or a derivative thereof, is versatile intermediate. They have active applications in organic synthesis for agrochemicals, pharmaceuticals, photographic, heat stabilizers, polymerization catalysts, flame-retardants, blowing agents for plastics, explosives, and dyes. Recently, hydrazine is applied to LCD (liquid crystal displays) as the fuel to make faster thin-film transistors. Hydrazone is a compound containing the group -NH·N:C-. It is formed from a condensation reaction aldehydes or ketones with hydrazines (commonly phenylhydrazine). It is used as an exotic fuel. Aromatic hydrazones are used to form indole by a ring closure reaction (Fischer synthesis). Hydrazones and hydrazines are converted to aldehydes and ketones to corresponding hydrocarbons by heating the carbonyl compound with sodium ethoxide (Wolf-Kishner reduction). Azide contains the group -N3 represented as a resonance hybrid of two structures, -N-N-��N+ �� -N=N+=N-. Organic azides are compounds replaced by a hydrocarbon group as in alkyl or aryl from hydrazoic acid (HN3)and have general formula RN3. Acyl azide is a compound in which the hydroxy group of a carboxylic acid is replaced by the azido group (HN3). Hydrazide is an acyl hydrazine. Acyl (-CO) is an organic radical formed by removal of a hydroxyl group from an organic acid (carboxyl group). Organic azides are useful for the synthesis of target compounds. They act as electrophiles on the nitrogen attached to the carbon and have electron-donating character for the neighboring carbon. | ||
| SALES SPECIFICATION | ||
|
50% SOLUTION |
||
|
APPEARANCE |
clear liquid | |
| CONTENT |
50.0%
min
|
|
|
AZIDE |
0.01% max |
|
|
SULFATE |
0.002% max |
|
| CHLORIDE |
0.005% max |
|
| IRON |
0.005% max |
|
| TRANSPORTATION | ||
| PACKING | 240kgs in drum | |
| HAZARD CLASS | 8 (Packing Group: III) | |
| UN NO. | 2922 | |
| OTHER INFORMATION | ||
| Hazard Symbols: C, Risk Phrases: 23/24/25-43-45-50/53 , Safety Phrases: 53-45-60-61 | ||
| GENERAL DESCRIPTION OF NITRIC ACID ITS SALTS | ||
| Nitric Acid is a colourless, highly corrosive, poisonous liquid (freezing point : -42° C, boiling point: 83° C) that will react with water or steam to produce heat and toxic, corrosive, and flammable vapors. It is toxic and can cause severe burns. It is an important industrial chemical for the manufacture of fertilizers, dyes, drugs, plastics, and explosives. Nitric acid is prepared commercially by the two-stage oxidation of ammonia (Ostwald process) to nitrogen dioxide. Ammonia gas is successively oxidized to nitric oxide and nitrogen dioxide by air or oxygen in the presence of a platinum gauze catalyst, which is then absorbed in water to form nitric acid. The resulting acid-in-water solution (about 50-78% by weight acid) can be dehydrated by distillation with sulfuric acid. It is miscible with water in all proportions. The nitric acid of commerce is typically a solution of 52% to 68% nitric acid in water. More concentrated solutions are available. It forms an azeotrope that has the composition 68% nitric acid and 32% water and that boils at 120.5°C. Solutions containing over 86% nitric acid are commonly called fuming nitric acid; they often have a reddish-brown color from dissolved nitrogen oxides.In aqueous solution it is both a strong acid and a powerful oxidizing agent. Among the many important reactions of nitric acid are: neutralization with ammonia to form ammonium nitrate, used widely in fertilizers and explosives; nitration of glycerol and toluene, forming the explosives nitroglycerin and trinitrotoluene, respectively; preparation of nitrocellulose; and oxidation of metals to the corresponding oxides or nitrates. Many compounds are oxidized by nitric acid. Nonmetallic elements such as carbon, iodine, phosphorus and sulfur are oxidized by concentrated nitric acid to their oxides or oxyacids with the formation of NO2 . Hydrochloric acid, aqueous HCl, is readily oxidized by concentrated nitric acid to chlorine and chlorine dioxide. The action of nitric acid on a metal usually results in reduction of the acid (i.e., a decrease in the oxidation state of the nitrogen). The products of the reaction are determined by the concentration of nitric acid, the metal involved (i.e., its reactivity), and the temperature. In most cases, a mixture of nitrogen oxides, nitrates, and other reduction products is formed. Relatively unreactive metals such as copper, silver, and lead reduce concentrated nitric acid primarily to NO2. The reaction of dilute nitric acid with copper produces NO, while more reactive metals, such as zinc and iron, react with dilute nitic acid to yield N2O. When extremely dilute nitric acid is used, either nitrogen gas or the ammonium ion (NH4+) may be formed. Nitrates, which are salts or esters of nitric acid contains NO3- (nitrate ion), are formed by replacing the hydrogen with a metal (e.g., sodium or potassium) or a radical (e.g., ammonium or ethyl). Some important inorganic nitrates are potassium nitrate (used in explosives, fireworks, matches, and fertilizers, and as a preservative in foods. It is sometimes used in medicine as a diuretic.), sodium nitrate (used in making potassium nitrate, fertilizers, and explosives.), silver nitrate (used in the preparation of silver salts for photography, in chemical analysis, in silver plating, in inks and hair dyes, and to silver mirrors.), and ammonium nitrate (Major uses are in fertilizers and explosives). Calcium nitrate is used in fertilizers; barium and strontium nitrates are used to color fireworks and signal flares; bismuth nitrate is used in making pharmaceuticals. Most nitrates are soluble in water, and a major use of nitric acid is to produce soluble metal nitrates. All nitrates decompose when heated and may do so explosively. The presence of nitrates in the soil is of great importance, since it is from these compounds that plants obtain the nitrogen necessary for their growth. Organic nitrates are esters formed by reaction of nitric acid with the hydroxyl (-OH) group in an alcohol. Nitrocellulose (or cellulose nitrate) is a highly flammable compound formed when cellulose materials are treated with concentrated nitric acid. The extent of the reaction between the cotton and acid can be varied to give a range of compounds, from the highly explosive gun-cotton to the flammable collodion cotton or pyroxilin. These are now used worldwide as propellants in cartridges and other ammunition. Collodion cotton and other less reactive forms of nitrocellulose are used chiefly in lacquers. They also form the basis of one of the earliest plastics, celluloid, made by the action on nitrocellulose of a solution of camphor in ethanol. Guncotton, fully nitrated cellulose, is used for explosives. Nitroglycerin is the nitric acid triester of glycerol and is more correctly called glycerol trinitrate. It is mixed with an absorbent material to form dynamite and is also used as a component of smokeless powder. Guncotton, fully nitrated cellulose is used for explosives. |
||